May 7, 2026
The US Food and Drug Administration (FDA) expects to replace the Manufacturer and User Facility Device Experience (MAUDE) database with its Adverse Event Monitoring System (AEMS) by the end of May 2026. We discuss what this means for safety data collection moving forward.
By Amelia Boldrick and Sarah Fitzgerald
As previously reported, the FDA intends to replace the MAUDE adverse event database with the unified regulated product adverse event database AEMS.
Background on MAUDE
The MAUDE database contains medical device reports (MDR) for reportable events that can be searched by keyword, exact phrase, brand name, UDI, model number, product code, manufacturer, report number, product problem, product class, event type, PMA/510(k) number, date range and exemption number. Manufacturers and other industry stakeholders have long used MAUDE to search and collect MDR data for a variety of activities including post-market surveillance, compiling clinical evaluation reports and/or identifying potential use errors in human factors engineering.
MAUDE is not without limitations. For example, only records from the previous ten years can be searched in the database, the searchable fields in MAUDE are only a fraction of the visible fields in an MDR public record, searchable fields in MAUDE may not always contain data and a single search can only return a maximum of 500 records.
An introduction to AEMS
While the name AEMS is new, the system itself is not. Better known by its former name, the FDA Adverse Event Reporting System (FAERS) until recently housed only drug, biologic, and cosmetic adverse event reports. When vaccine adverse event records were added from the VAERS system and event reports for all current product types began to be processed in real time, FAERS was renamed to AEMS. Interestingly the FAQ linked in the AEMS dashboard also still refers to the system as FAERS at the time of this update.
What to expect in AEMS when medical device reports are added
As with the migration of vaccine data from VAERS to AEMS, historical medical device data will be migrated and new MDRs will be processed in AEMS in real time. Although the FDA considers AEMS to be intuitive to navigate, those familiar only with searching in MAUDE may find the AEMS dashboard radically different in appearance and search functionality.
Depending on product type and report format for the regulated products currently available in AEMS, different report fields are displayed:
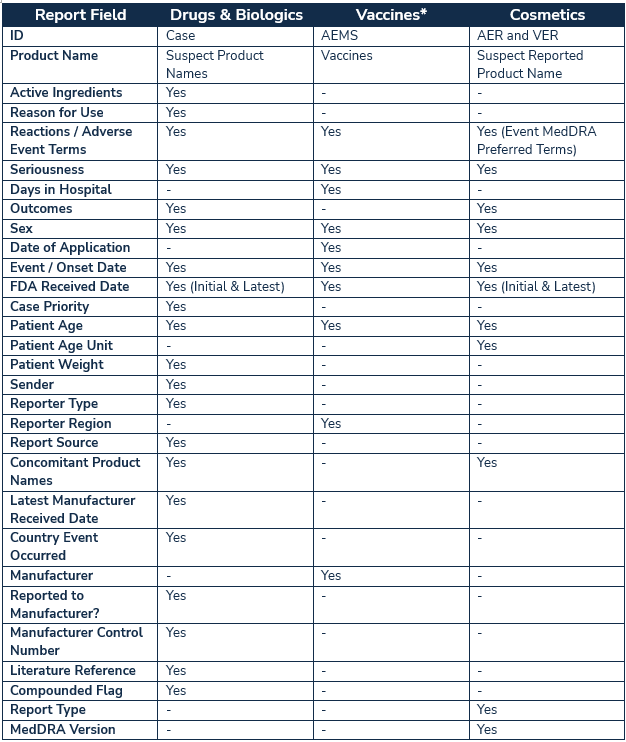
* Vaccine event cases can be viewed individually by AEMS ID to obtain additional event data
Concluding remarks
The AEMS currently manages different sets of data from different types of adverse event report for different types of regulated products. There should be no difficulty searching AEMS and exporting search results as is currently done in MAUDE.
Emergo by UL experts look forward to sharing more information about the medical device MDRs in AEMS.
Request more information from our specialists
Thanks for your interest in our products and services. Let's collect some information so we can connect you with the right person.







